
Ling Zhang, Zebiao Qiu, Ruijuan Wen, Pan He, Hong Tian, Lingya Peng*, Rui Cao, Haonan Peng*, Yu Fang. Chem. Commun., 2026, 62, 5739-5743. DOI: 10.1039/d5cc07311a
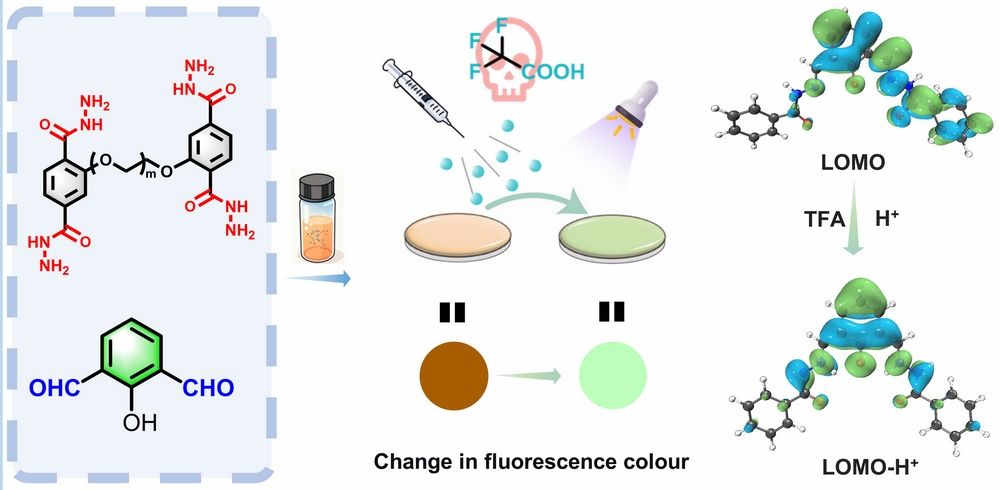
Manipulation of intramolecular charge transfer (ICT) is a powerful tool for tuning the photophysical properties of conjugated materials, which holds profound significance for fluorescent sensing applications. In donor-π-acceptor (D-π-A) systems, ICT refers to the electronic redistribution within a molecule upon excitation, where the electron density shifts between the donor and acceptor units, thereby resulting in fluorescence emission. However, in most cases, protonation of such systems disrupts the ICT balance, enhances intramolecular charge separation, and activates non-radiative decay channels (e.g., C=N isomerization and backbone rotation), ultimately leading to fluorescence quenching rather than enhancement. This "protonation-ICT-quenching" mechanism has been widely exploited in the field of sensing, but it also limits our ability to predictably regulate the emission color and efficiency, especially in solid films where conformational freedom and microenvironmental heterogeneity are more difficult to control, making this limitation more pronounced.
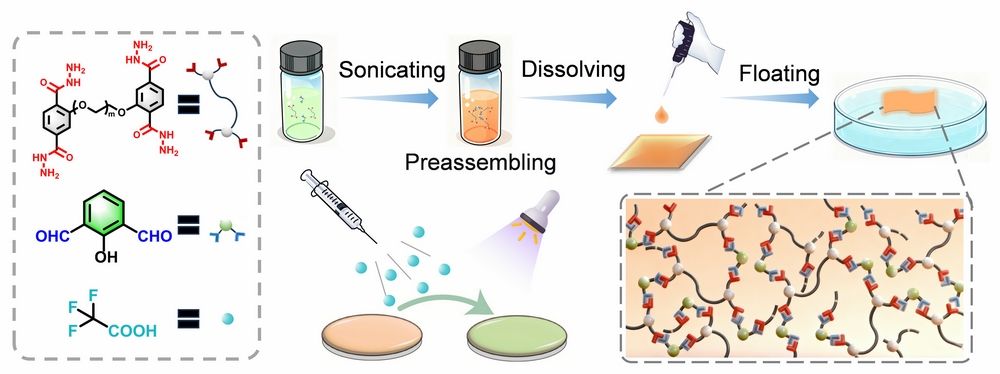
Figure 1. Schematic of the nanofilm fabrication process, depicting precursor solution preparation, drop-casting, interfacial reaction, and fluorescence changes before and after TFA exposure.
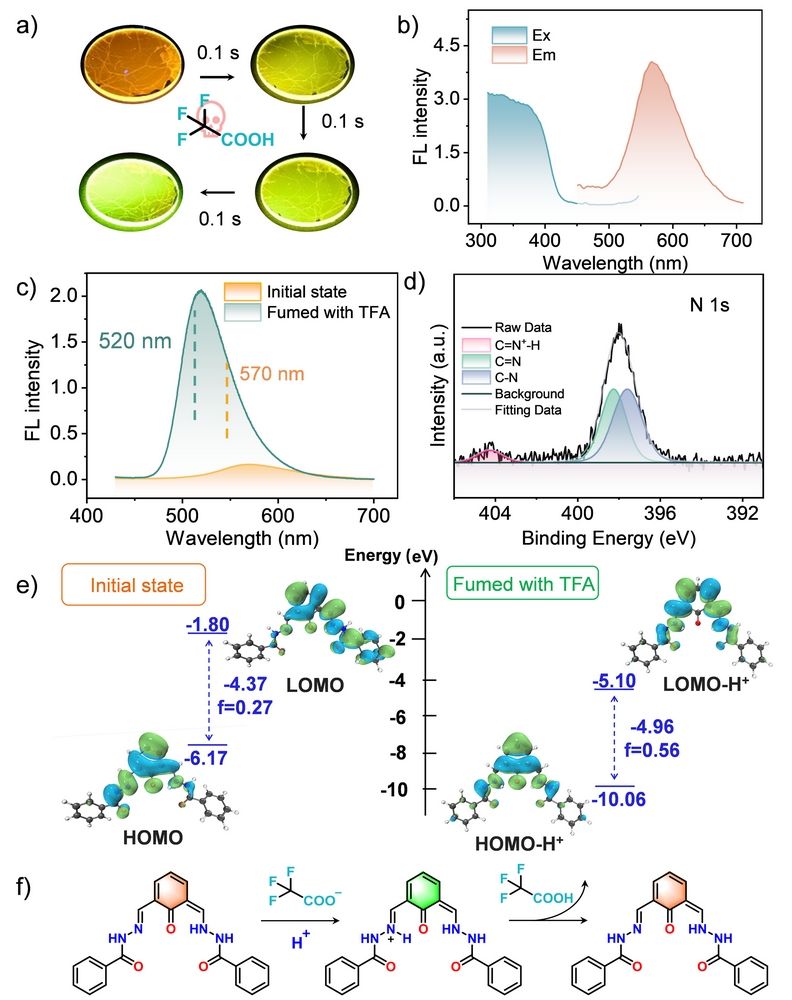
Figure 2. (a) Fluorescence response of the DTH–DFP nanofilm to saturated TFA vapour under 365 nm UV light. (b) Nanofilm fluorescence spectra: excitation (cyan) and emission (orange). (c) Fluorescence emission spectra before and after exposure to saturated TFA vapour. (d) N 1s XPS spectrum after TFA treatment. (e) Optimised HOMO–LUMO orbital diagrams of the repeating unit within the nanofilm before and after protonation. (f) Proposed mechanism of binding of the nanofilm to TFA.
In this work, we embedded protonation sites into charge-transfer active segments to promote electron rearrangement of the surrounding framework, while utilizing the two-dimensional confinement effect to suppress non-radiative decay, thus constructing a turn-on strong light-up fluorescent nanofilm. By constructing a highly crosslinked acylhydrazone bond network based on 2-hydroxyisophthalaldehyde and acylhydrazone-linked oligomers as building blocks at the air/dimethyl sulfoxide (DMSO) interface, we successfully fabricated the DTH-DFP nanofilm. The proton-sensitive Ph-C=N-N-C=O segment in the nanofilm enables precise regulation of intramolecular charge transfer properties; site-selective protonation at the imine nitrogen, combined with the two-dimensional interfacial confinement effect, can induce intramolecular electron redistribution, reduction of conjugated area, increased HOMO-LUMO orbital overlap, and enhanced oscillator strength, ultimately achieving blue-shifted fluorescence turn-on. This provides a rapid, and sensitive detection platform for trifluoroacetic acid (TFA) vapor sensing (limit of detection, LOD = 1.48 ppm).
First Author: Zhang Ling, master’s student, Shaanxi Normal University
Correspondence Authors: Prof. Peng Haonan and Dr. Peng Lingya, Shaanxi Normal University
Full Text Link: https://doi.org/10.1039/D5CC07311A
 Latest Updates
Latest Updates






