
Jiman He, Xingwang Chen, Jiahui Peng, Mengyu Wang, Sensen Qian, Shujiang Ding, Hongyang Zhao*, Hongtao Bian*. ACS Appl. Mater. Interfaces, DOI: 10.1021/acsami.5c25987
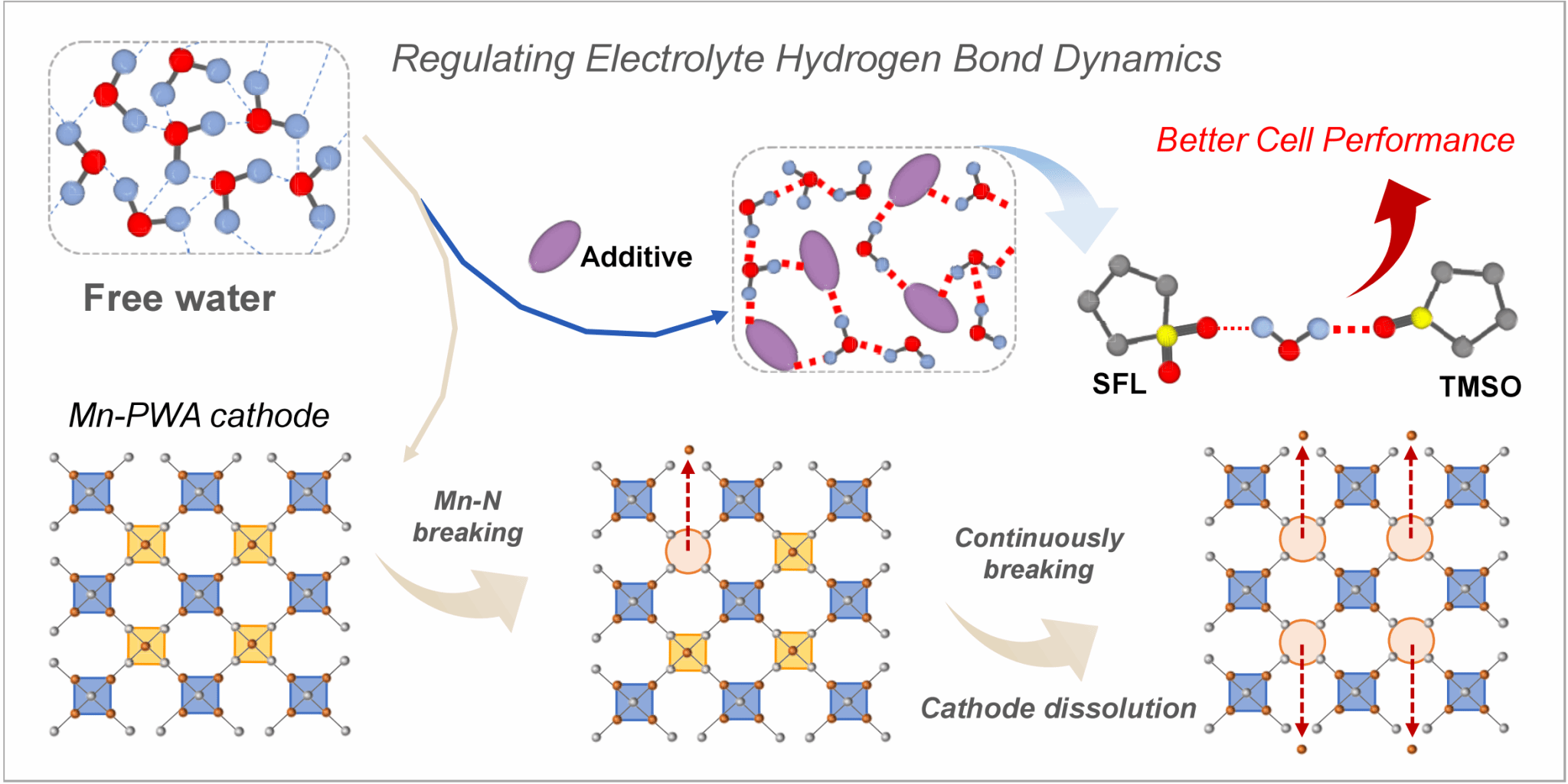
Aqueous sodium-ion batteries hold great promise for large-scale energy storage due to their high safety, low cost, and environmental friendliness. However, the high reactivity of water molecules at the electrode interface continuously induces the dissolution of cathode materials; in particular, Prussian white-type cathodes are more prone to structural degradation during long-term cycling, which severely limits their practical application. Previous studies have largely attributed material instability to the thermodynamic activity of water; however, this research demonstrates that the true determinant of cathode stability lies not only in “how reactive the water is,” but also in the dynamic behavior of the hydrogen-bond network formed by water molecules at the interface.
To elucidate this mechanism, the research team selected two cyclic sulfoxide molecules with similar structures but different sulfur oxidation states as model additives: tetramethylene sulfoxide (TMSO) and cyclobutene sulfoxide (SFL). Combining ultrafast infrared spectroscopy, steady-state infrared spectroscopy, and density functional theory calculations, they systematically compared their effects on the hydrogen-bonding structure and dynamics of water molecules at the molecular level. The study found that TMSO, due to its lower sulfur oxidation state, possesses a higher electron density in its S=O group and exhibits stronger hydrogen-bonding acceptor capabilities. It forms strong, single-bond hydrogen bonds with water molecules, stabilizing the local water network while only moderately slowing down the reorientation dynamics of water molecules; In contrast, SFL, due to its dual S=O structure, tends to form polycoordinated hydration configurations, resulting in an overly rigid local hydrogen-bond network.
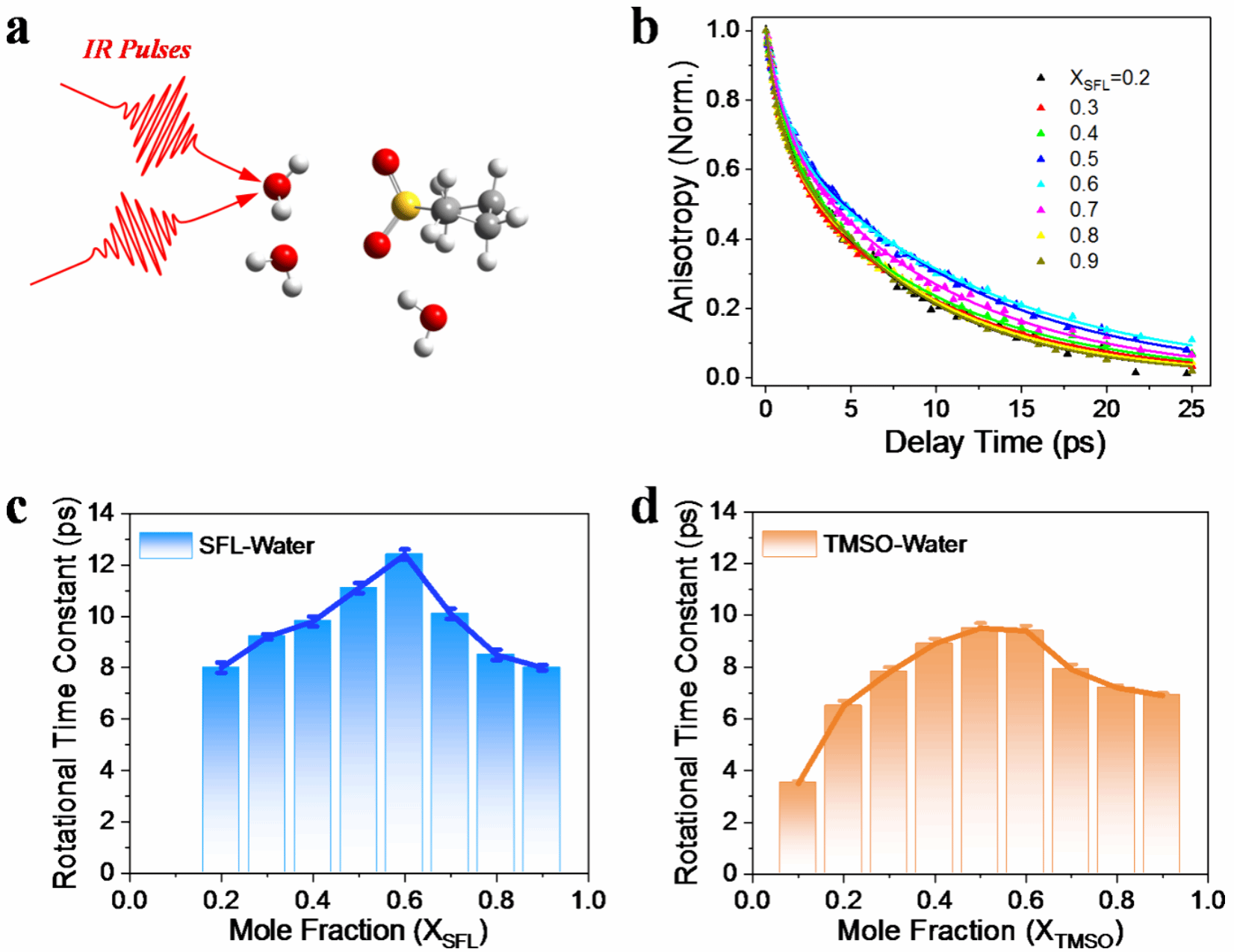
Fig. 1. Ultrafast infrared spectroscopic characterization of hydrogen-bond dynamics of water molecules in SFL/ H2O and TMSO/ H2O mixtures at different concentrations.
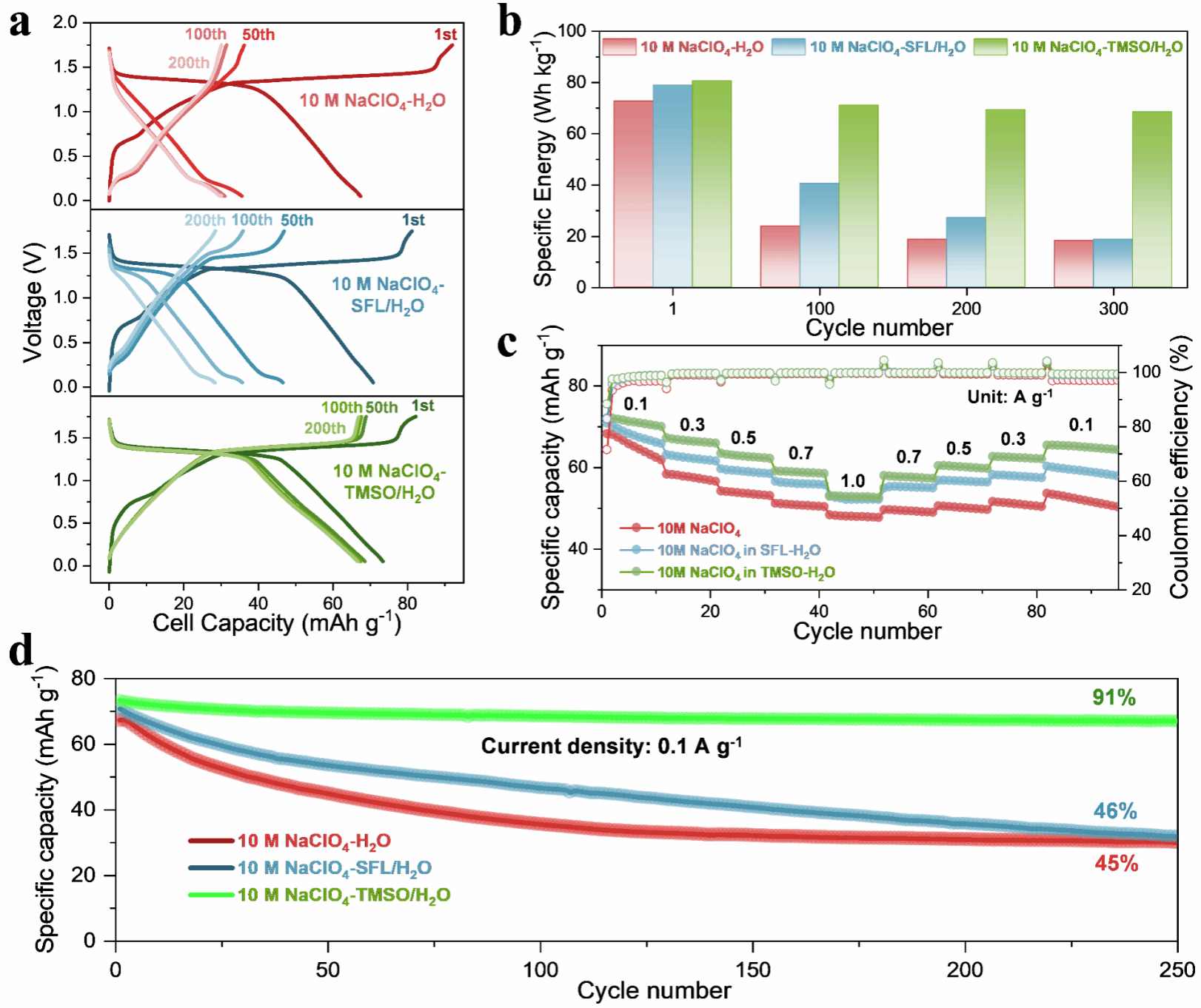
Fig. 2. Electrochemical performance of Mn-PWA/NTP full cells with different electrolytes.
Ultrafast IR results further indicate that TMSO’s regulation of water molecule hydrogen-bonding dynamics exhibits a “balanced” characteristic: it suppresses the erosion of the cathode framework by active water at the interface without sacrificing the structural flexibility required for ion transport due to an overly rigid network. Based on this understanding, the research team introduced TMSO into a high-concentration NaClO4 aqueous electrolyte for the protection of the manganese-based Prussian white (Mn-PWA) cathode. Electrochemical test results show that the TMSO-modified electrolyte effectively suppresses the leaching of transition metals such as Fe and Mn, significantly improving the cycling stability of the full cell: the Mn-PWA/NTP full cell achieved a capacity retention rate of 91% after 250 cycles at 0.1 A·g-1; In further pouch cell testing, the battery retained 89% of its initial capacity after 150 cycles, demonstrating excellent application potential. The paper also demonstrates the system’s stable performance under longer cycling conditions.
This study is the first to directly link the picosecond-scale hydrogen-bonding dynamics of water molecules with macroscopic battery cycling stability. It establishes a clear connection between “molecular electronic structure—hydrogen-bonding geometry and dynamics—interface stability—device performance” and proposes design principles for optimizing interfacial water behavior by regulating the electronic structure of electrolyte molecules.
These findings not only provide new insights for protecting the cathode in highly stable aqueous sodium-ion batteries but also offer a theoretical foundation and molecular design strategies applicable to other electrochemical systems—such as zinc-ion batteries and electrocatalysis—that rely on interfacial water stability.
First Authors: He Jiman, Master’s student, Shaanxi Normal University; Chen Xingwang, Master’s student, Xi’an Jiaotong University; Peng Jiahui, Master’s student, Shaanxi Normal University
Correspondence Authors: Prof. Bian Hongtao, Shaanxi Normal University; Assoc. Prof. Zhao Hongyang, Xi’an Jiaotong University
Full Text Link: https://pubs.acs.org/doi/10.1021/acsami.5c25987
 Latest Updates
Latest Updates






