
Jing Li#, Xubin Wang#, Jiahai Chen, Zhouyu Chen, Simin Lin, Rong Miao* and Yu Fang*. CCS Chem. 2026, DOI: 10.31635/ccschem.026.202607606
The photophysical properties of organic luminescent materials are strongly influenced by molecular conformation, intramolecular motion, and solid-state packing. In aggregation-induced emission (AIE) systems, restricting intramolecular rotations and vibrations is particularly important because it helps suppress nonradiative decay and improve emission efficiency. Dimeric systems, with their well-defined structures and clear interchromophore interactions, have therefore been widely used as model systems for understanding conformation-property relationships. However, conventional linkers, such as flexible chains, rigid aryl groups, and macrocycles, can only partially regulate chromophore distance and coupling, and often do not provide sufficient control over three-dimensional molecular conformation. In this context, [5]helicene is especially attractive because of its rigid, chiral, and sterically demanding structure. It can act not only as a linker, but also as a conformational editor, offering a useful strategy for tuning both luminescence and chiroptical properties.
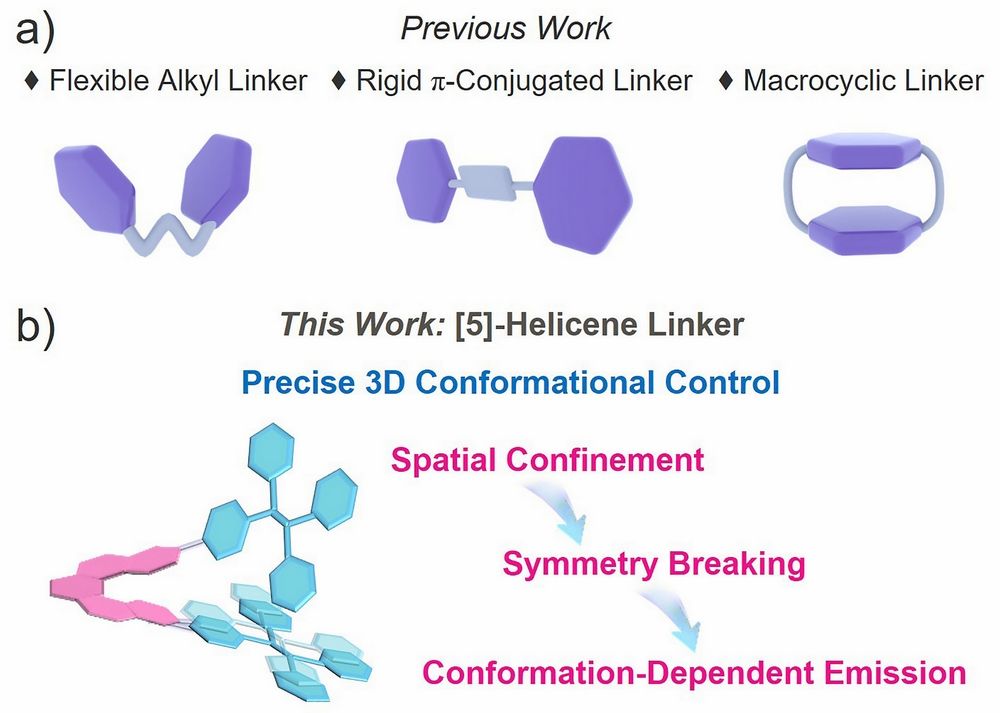
Figure 1. Schematic comparison of traditional linker-based dimer design strategies (a) with the spatial-confinement-driven structural symmetry-breaking strategy introduced in this work (b).
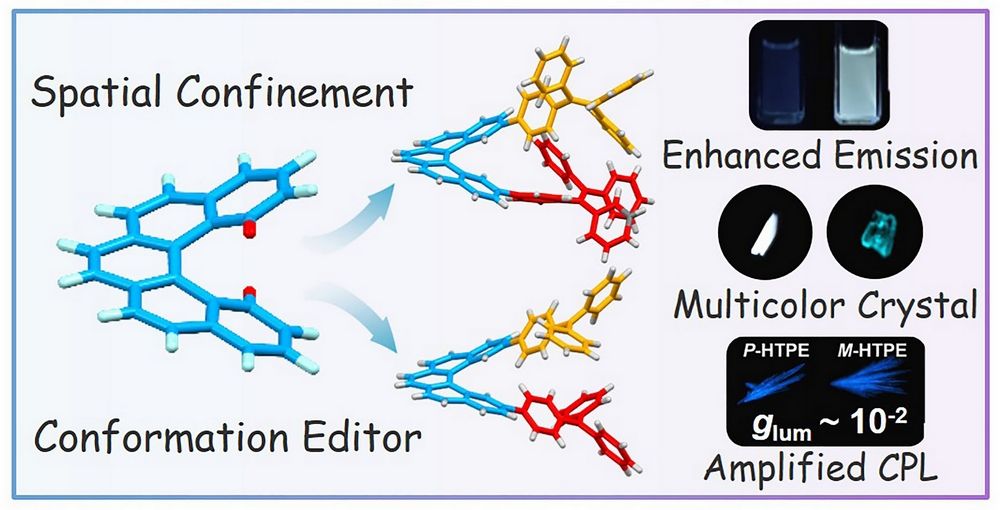
In this work, a rigid chiral [5]helicene unit was used to construct two dimers, HTPE and HTPA, containing tetraphenylethylene (TPE) and triphenylamine (TPA), respectively. The purpose was to regulate the conformation and luminescence behavior of AIE-active molecules through spatial confinement. The results show that [5]helicene does more than simply connect two luminophores. By restricting intramolecular motion, it greatly enhances fluorescence in solution: HTPE is 47 times more emissive than TPE, while HTPA shows a 154-fold enhancement compared with TPA. At the same time, the confined three-dimensional environment created by the helicene linker leads to different locking effects for luminophores of different sizes. In HTPE, the bulkier TPE units favor an asymmetric conformation, whereas in HTPA, the smaller TPA units adopt a symmetric arrangement. Single-crystal analysis and theoretical calculations further show that the asymmetric conformation of HTPE is thermodynamically preferred. This conformational locking also gives rise to different polymorphs with tunable fluorescence. In addition, the chirality of the helicene unit endows HTPE with clear circular dichroism and circularly polarized luminescence in crystals, aggregates, and doped films, with a glum value reaching the 10-2 level in the crystalline state. HTPE can also serve as a chiral donor in a circularly polarized Förster resonance energy transfer (C-FRET) process, transferring both energy and chirality to acceptor dyes such as rhodamine B and thereby enabling multicolor circularly polarized luminescence. Overall, this work provides a useful approach for regulating the fluorescence and chiroptical properties of multichromophoric systems through helical linkers.
First Authors: Li Jing and Wang Xubin, doctoral candidates, Shaanxi Normal University
Correspondence Authors: Prof. Fang Yu, A/Prof. Miao Rong, Shaanxi Normal University
Full Text Link: https://www.chinesechemsoc.org/doi/pdf/10.31635/ccschem.026.202607606
 Latest Updates
Latest Updates






