
Xinyu Gou, Zhaolong Wang, Ke Liu, Qiyuan Shi, Xue Gu, Taihong Liu*, Yu Fang*. Sens. Actuator B-Chem., 2024, 399, 134857.
Chlorine dioxide (ClO2), as a green disinfectant, is widely used due to outstanding performance and the resultant of chloride ions, not the carcinogenic trihalomethanes formed by the typical disinfectants (NaClO). Typically, ClO2 is used at a concentration level between 0.1 and 5 mg/L for water treatment, therefore, development of simple, sensitive, and selective identification method of ClO2 used in solution phase is important. Over the past few decades, GC-MS, UV-vis spectrophotometry, electrochemical methods, semiconductor sensors, and resonance scattering techniques have been developed. The approaches, however, may suffer from some shortcomings, such as complicated operation, time-consuming, and high-power consumption, etc. In contrast, fluorescence techniques feature the merits of high sensitivity, fast response, and great designability. Therefore, we hope to develop new agent for high-performance fluorescence detection of ClO2 in situ and real-time.
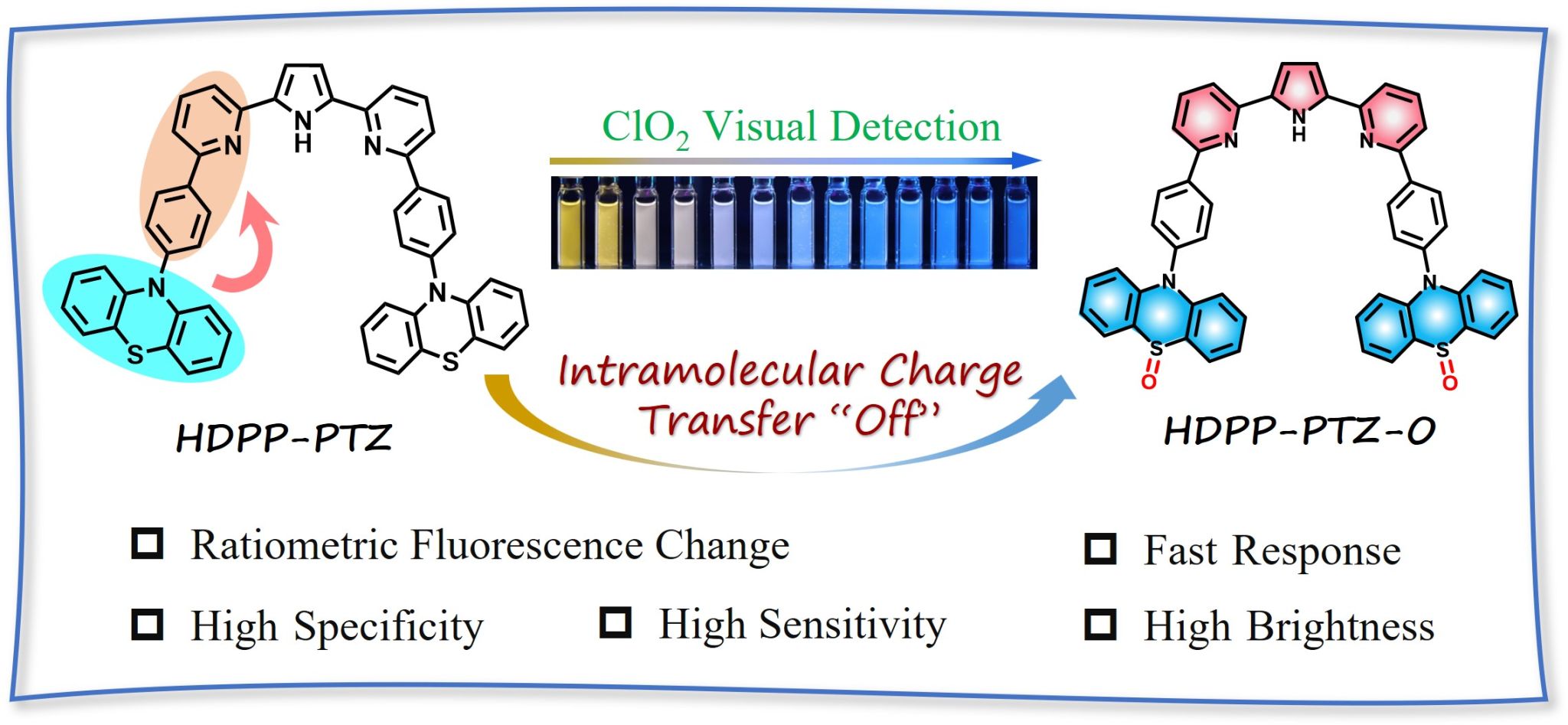
Figure 1. Photographs of HDPP-PTZ toward various concentrations of ClO2 in THF/water (1/1, v/v) and the possible sensing mechanism.
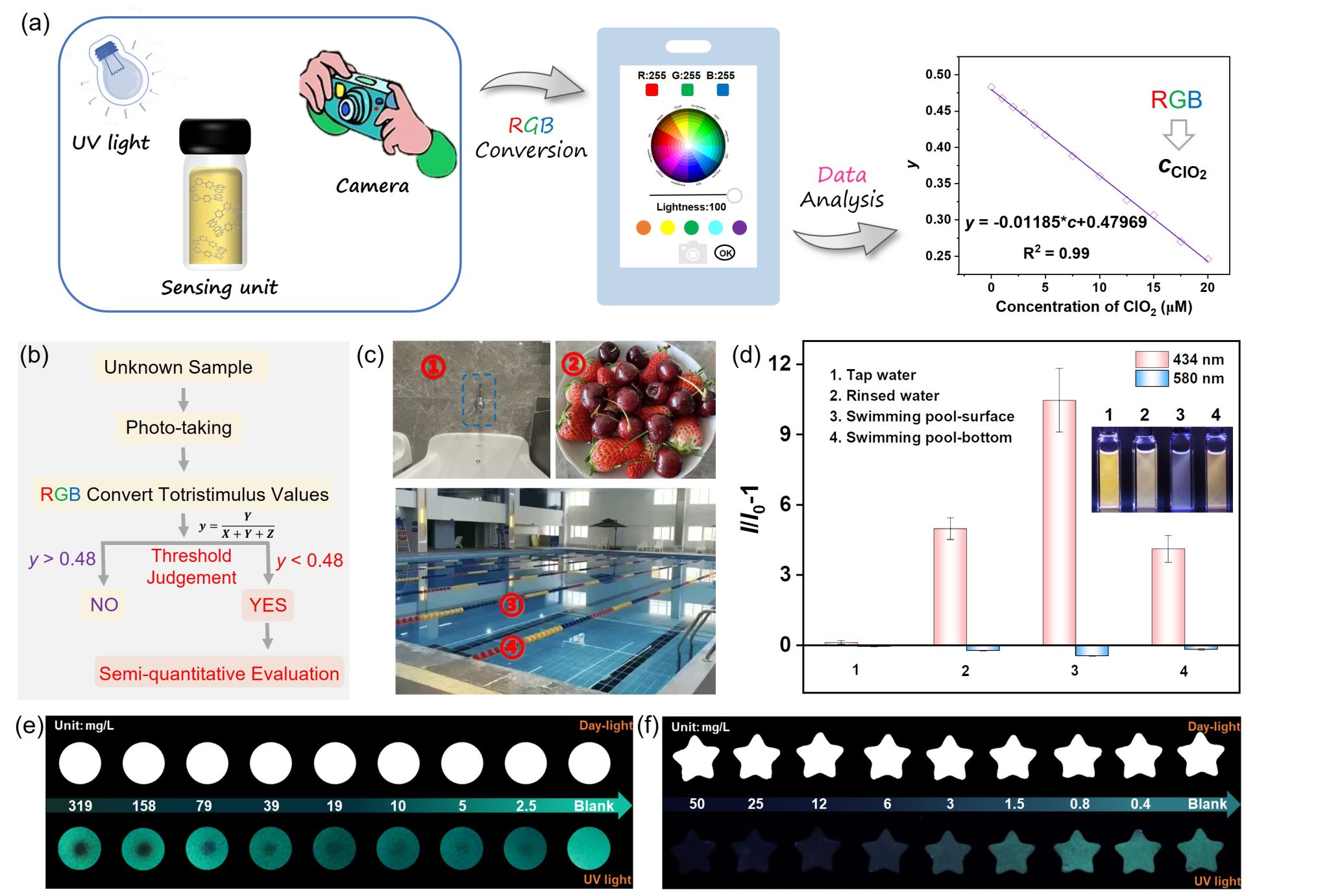
Figure 2. (a) Schematic illustration of RGB analysis to evaluate the content of ClO2 using the established calibration equation. (b) Schematic illustration of the processes of sample evaluation in field applications. (c) Pictures of the ClO2 detection in the simulation scenes. (d) Representative sensing performance of the fluorescence probe at 434 and 580 nm, each error bar was calculated from three parallel tests. Inset showed the pictures of the fluorescence probe HDPP-PTZ reacted with real samples. Photographs of HDPP-PTZ coated strips toward detecting aqueous and gaseous ClO2 (e, f) under day-light (upper) and UV light (lower).
Phenothiazine (PTZ) as a strong electron-donating is often used as an active fluorescence component in push-pull chromophores. The sulfur atom (+2) of PTZ can be oxidized to sulfoxide (+4) or sulfoxide (+6), which not only introduces d-pπ bonds to diminish the electron delocalization throughout the conjugation skeleton, but also results in drastic change in the photo-electronic properties of PTZ from a strong donor to a weak acceptor. Herein, we designed the PTZ triad (HDPP-PTZ) with the properties of intramolecular charge transfer (ICT). Dual emission of the compound in solution assigned as LE and ICT emission. The polarity sensitive ICT emission of HDPP-PTZ fluorophore totally disappeared upon oxidizing the sulfur atom in PTZ unit into sulfoxide. Inspired by the discovery, a highly selective, sensitive and rapid detection method for a widely used disinfectant chlorine dioxide (ClO2) is established. The detection limit (DL) of ClO2 in THF/ water is < 0.052 mg/L, satisfying the setting limit by WHO (1.0 mg/L), the response time is less than 20 s and no significant interference. Take advantage of the remarkable fluorescence changes, real samples are visualized and evaluated by RGB analysis. Testing strips are further fabricated for in-situ and at real-time detection of ClO2 both in aqueous solution and gas states. Present study provides significant understanding of the oxidation states of the PTZ derivatives and guides development of fluorescent probes for specific analyte.
First Author: Doctoral candidate Gou Xinyu, Shaanxi Normal University
Correspondence Authors: Prof. Fang Yu and Assoc. Prof. Liu Taihong, Shaanxi Normal University
Full Text Link: https://doi.org/10.1016/j.snb.2023.134857