
Zanru Tan, Jiang Feng, Jiangping Liu,* Taihong Liu,* Huihui Wu*, Hui Chao*. Dalton Trans., 2025, DOI: 10.1039/D4DT03426K.
PDT has emerged as a promising alternative in recent decades. In a PDT regimen, a photosensitizer (PS) absorbs light energy and transits to the excited state by which this energy (type II ) or the excited electron (type I) is further shifted to the neighboring oxygen molecules, generating reactive oxygen species (ROS). These ROS oxidize pivotal macro-biomolecules, such as lipids, proteins, and DNA, triggering deeper biological responses and ultimately inducing cell death. Nevertheless, current commercially available PSs, such as porphyrin and chlorin derivatives, suffer from various limitations, including tedious preparation, poor photostability, and inadequate selectivity. Therefore, a novel class of PSs with bold structural novelty is highly demanded to push the envelope of this field.
Many Ir(III) complexes have been considered candidate drugs for anticancer therapy due to their favorable photophysical attributes and unique physiological properties, including strong photostability, ease of synthesis, and modifications. Besides, phosphorescent cyclometalated Ir(III) complexes often specifically accumulate in various cellular compartments such as mitochondria, endoplasmic reticulum, lysosomes, Golgi apparatus, and nuclei, providing a convenient way to induce cytotoxicity through the generation of ROS. Whilst mononuclear Ir(III) complexes have been extensively used as PSs for anticancer therapy, their dinuclear counterparts remain largely underexplored. A recent study suggested that a dinuclear cyclometalated Ir(III) complex in which the monomeric units were simply bridged by a flexible alkyl chain outperformed its mononuclear form in both photoluminescence quantum yield and singlet oxygen quantum yield. Indeed, homonuclear bimetallic complexes are often paired with interesting properties that are beyond the reach of their monomers. These advances prompt us to scrutinize the optical properties of a novel series of dinuclear Ir(III) complexes with rigid bridging ligands in which the optimized planar configuration may stand a chance to largely reshape its 2PA transition probabilities. The 2PA process involves the simultaneous absorption of two photons. Compared to single-photon absorption, 2PA offers an elegant way to excite Ir(III) complexes in the near-infrared regime and allows deeper tissue penetration for PDT applications.
Herein, a series of novel binuclear Ir(III) complexes were synthesized. In contrast to the routine multinuclear framework, the Ir(III) monomers were interfaced from the flank in an asymmetric manner via two large conjugate planes. Their photophysical properties and photocytotoxicity were investigated. Interestingly, benefiting from the potentially extended planar configuration, the dinuclear complexes feature significant enhancement in 2PA absorption compared to the reported mononuclear complexes.
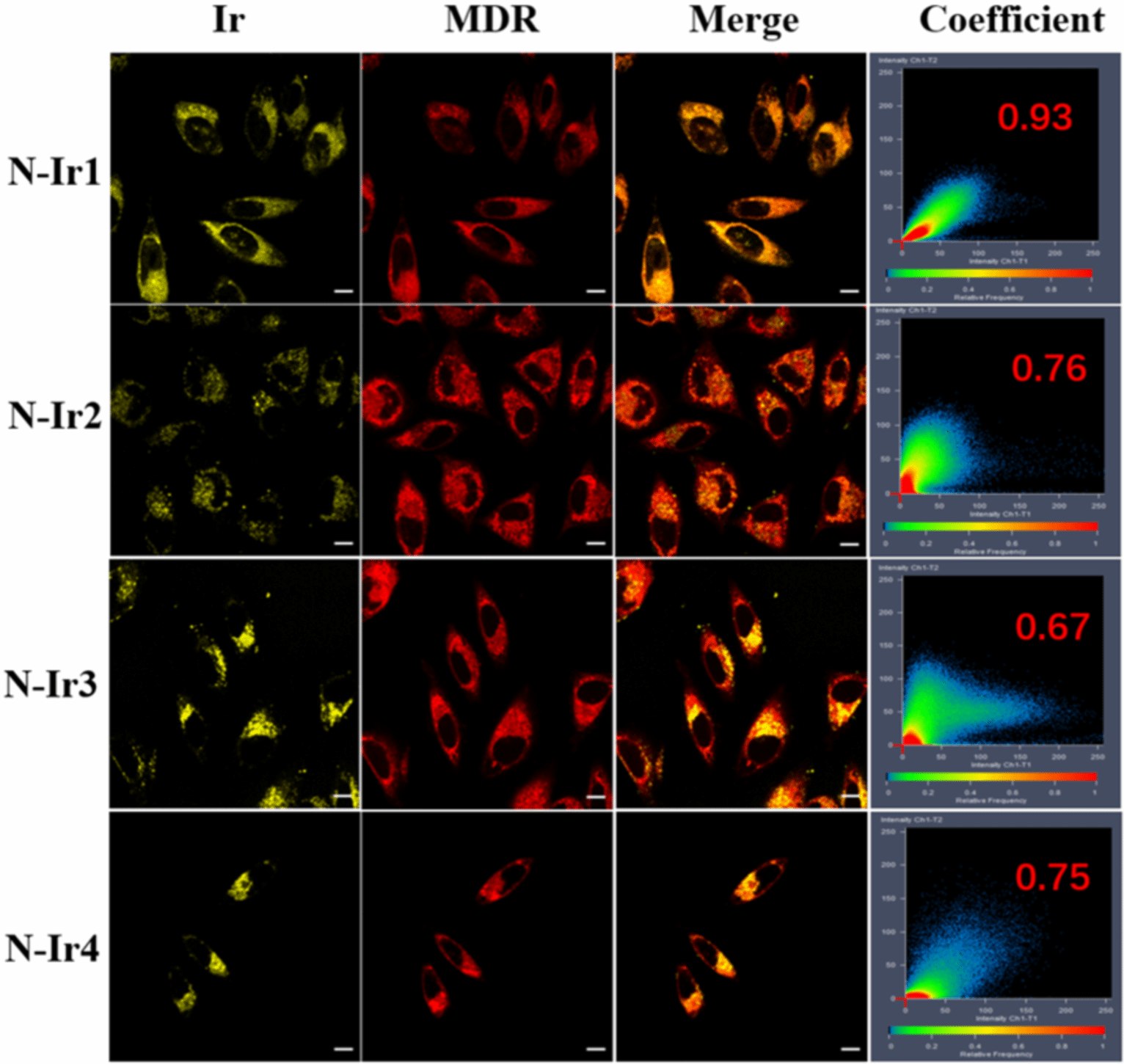
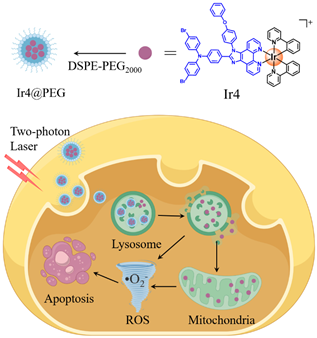
Figure 1. Chemical structures of the Ir(III) complexes, the putative action mechanism in 2PA PDT and CLSM imaging of A375 cells incubated with the Ir(III) complexes
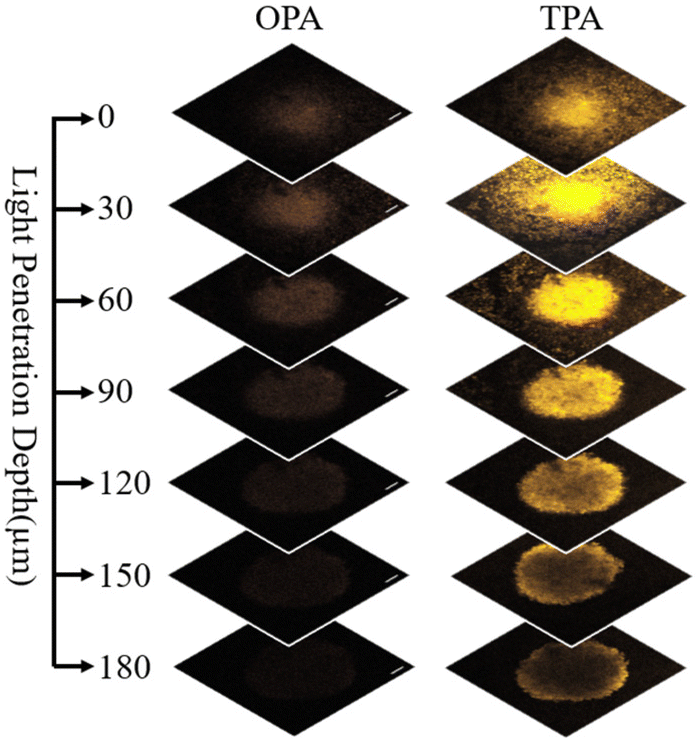
Figure 2. Penetration depth comparasion of the complexe N-Ir4 in 3D multi-cellular tumor spheroids upon one- or two-photon irradiation. The excitation wavelengths of OPM and TPM were 405 nm and 720 nm, respectively
First and second Author: Tan Zanru, doctoral candidate, Sun Yat-sen University; Feng Jiang, master’s candidate, Shaanxi Normal University
Co-Correspondence Authors: A/Prof. Liu Taihong, Shaanxi Normal University; Prof. Chao Hui, Sun Yat-sen University.
Full Text Link: https://doi.org/10.1039/D4DT03426K