
Linxuan Huang, Xinxiang Gao, Xinyao Mao, Ishfaq Ullah, Zhijie Zhou, Ejaz Hussain,* Yinghui Xiao, Haonan Peng,* and Yu Fang. Macromol. Rapid. Commun. 2025. DOI: 10.1002/marc.202401048
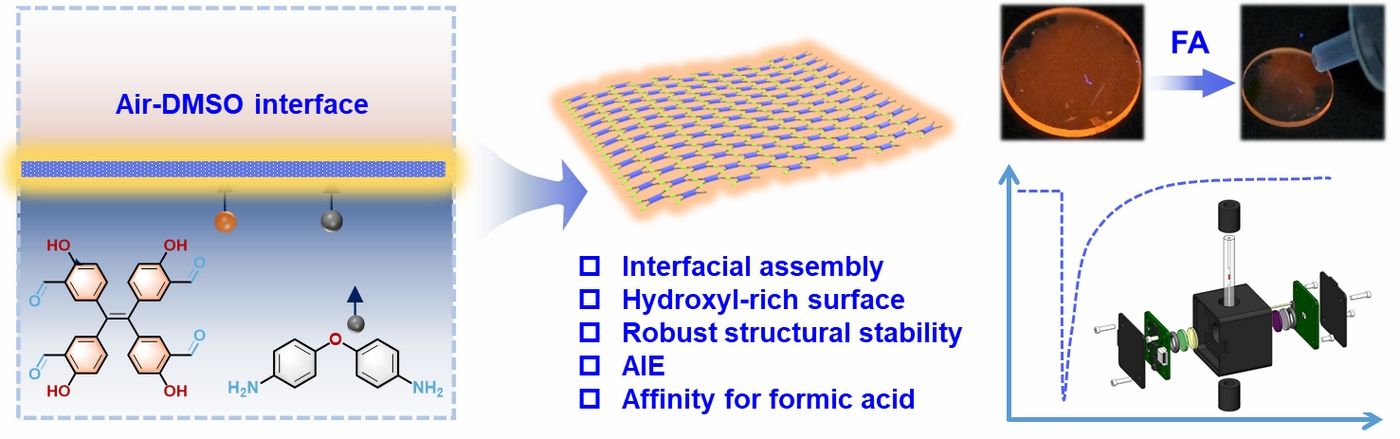
Formic acid (FA) is widely used in the chemical industry but also has significant impacts on air pollution, global acidification, and environmental degradation. Due to its corrosiveness and potential health hazards, including respiratory issues and skin burns, sensitive and real-time FA detection technologies are needed to allow for timely mitigation measures and ensure compliance with relevant regulations. Film-based fluorescent sensors are considered an important technology for detecting volatile organic compounds (VOCs), offering high sensitivity, intuitive visual signal output, and the potential for real-time on-site monitoring. Among them, organic molecule-based fluorescent sensors are particularly notable due to their structural versatility, tunable optical properties, and ease of functionalization to selectively detect specific analytes. In this work, a polymer nanofilm was synthesized through the dynamic condensation of a hydroxyl-modified tetraphenylethylene derivative with 4,4′-diaminodiphenyl ether at the air-liquid interface. The resulting nanofilm exhibits excellent luminescent properties, flexibility, and tunable surface morphology. Its bright fluorescence is primarily attributed to aggregation-induced emission (AIE) properties and the rich hydrogen bonding network within the film structure. The sensor fabricated from this nanofilm achieves an ultra-low detection limit of 550 ppt for formic acid vapor, a rapid response time of 0.3 s, and excellent reversibility. It also demonstrates higher selectivity for formic acid over other VOCs, making it ideal for real-time monitoring. Additionally, the nanofilm's multimodal sensing capabilities, including responses to HCl and NH3, highlight its potential for diverse environmental and industrial applications.
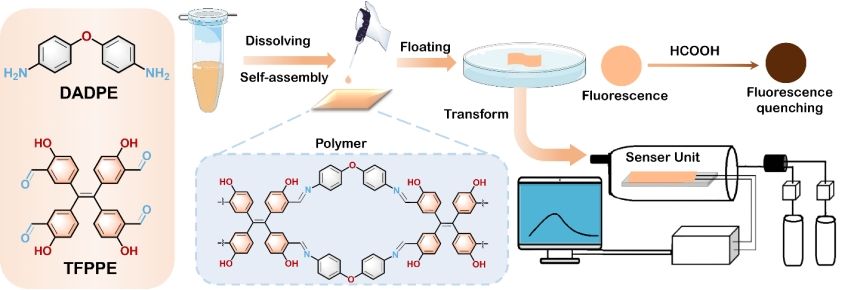
Figure 1. Illustration of the chemical structures of the precursor and linker, the process of polymer nanofilm fabrication, integration into a functional formic acid detection system, and its fluorescence quenching behavior in response to formic acid.
The mechanism of formic acid sensing by the fluorescent nanofilm was investigated using techniques such as NMR and fluorescence lifetime measurements. Formic acid molecules interact with the imine bonds in the nanofilm, forming weak hydrogen bonds that disrupt the hydrogen bonding network. This interaction alters the electronic environment of the imine groups, blocks proton donors (─OH), and promotes non-radiative decay pathways, leading to fluorescence quenching.
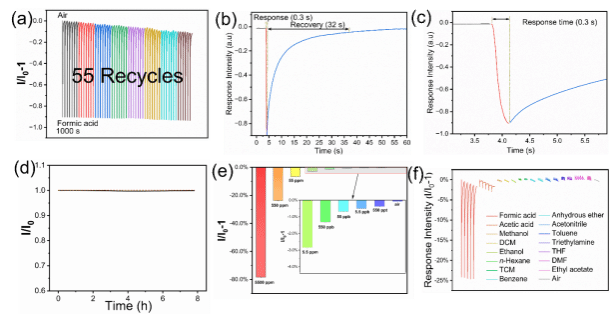
Figure 2. Sensor performance studies: a) Reproducibility of the sensor response. b) Recovery and response time. c) Expanded graph of response time. d) Photochemical stability e) sensitivity and, f) selectivity of fluorescent nanofilm. (λex = 440 nm, and λem = 620 nm).
First Authors: Huang Linxuan and Gao Xinxiang, master’s students, Shaanxi Normal University
Correspondence Authors: Prof. Peng Haonan and Dr. Ejaz Hussain, Shaanxi Normal University
Full Text Link: https://doi.org/10.1002/marc.202401048