
Haixia Chang, Jiang Feng, Xin-Ao Liu, Rong Miao,* Taihong Liu,* Liping Ding, and Yu Fang. Chem. Sci., 2025, DOI: 10.1039/D5SC00013K
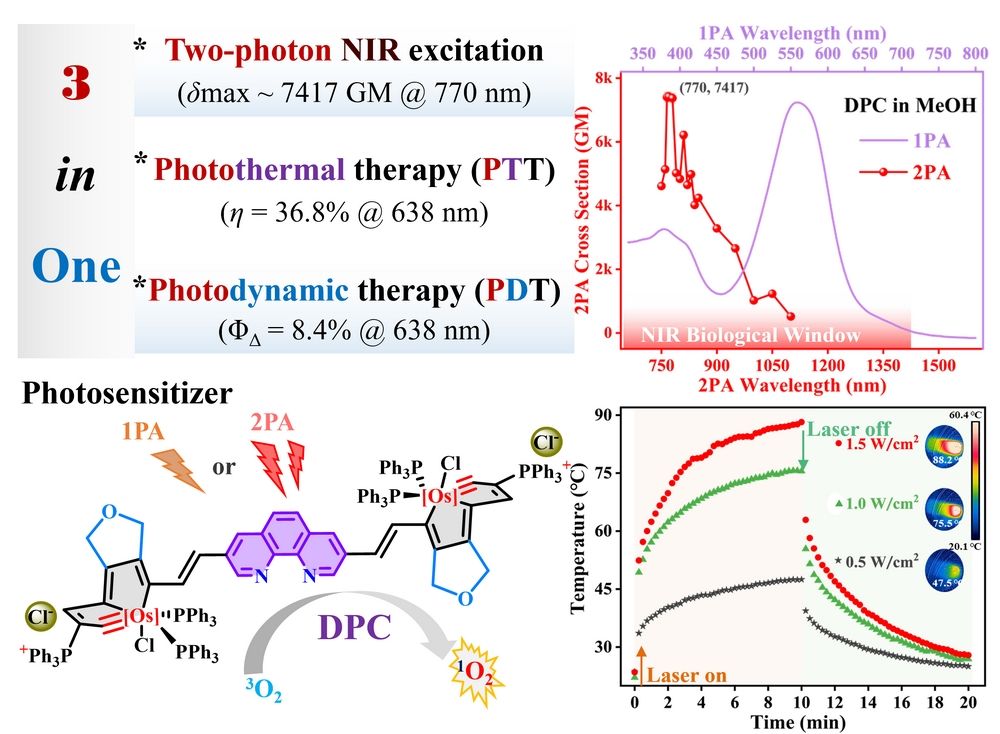
Phototheranostics has been evoked as an emerging and promising approach toward cancer therapy because of the advantages such as minimal invasiveness, spatiotemporal accuracy, and precise controllability. The non-destructive photodynamic (PDT) and photothermal (PTT) therapies depend on photosensitizers that produce heat or reactive oxygen species when irradiated to destroy cancer cells. Compared to one-photon absorbing photosensitizers, two-photon exciting materials in the significant near-infrared (NIR) biological spectral window (650 ~ 1450 nm) show distinct advantages of deeper tissue penetration, minimal light loss, and improved phototheranostic properties. Two-photon absorption (2PA) enables the excitation of photosensitizers by longer spectral region with lower energy and allows higher spatial resolution via quadratic dependence of the 2PA rate on the incident laser light intensity. Searching for photosensitizers possessing large 2PA cross section (δ2PA), together with exceptional photostability, good biocompatibility, and high therapeutic efficacy remains a significant challenge in material science and biomedicine. The metallaaromatic chemistry has recently evoked considerable attention due to their Möbius and adaptive aromaticity, diverse reactivity, excellent thermodynamic stability, and outstanding nonlinear optical performances. Understanding their excited-state dynamics is crucial for manipulating their photophysical properties and developing related optoelectronic and phototheranostics materials.
Herein, we compared the linear photophysical behaviors and 2PA properties of dipole SPC and quadrupole DPC chromophores. The excited-state dynamics of quadrupole double-phenanthroline-carbolong DPC were focused and elucidated properly. Its significant 2PA properties within the spectral range of 750 ~ 1100 nm were investigated using the open-aperture Z-scan instrument. Of particular interest on the excited-state photophysical dynamics, nonradiative relaxations and the intramolecular charge transfer (ICT) in DPC were comprehensively investigated. Ultrafast transient absorption spectra clearly unveiled the excited-state dynamics of DPC, including the originally occupied Franck−Condon state, rapid ICT, and ISC processes to the T1 state. The long T1 state lifetime was evident from the stable excited-state absorption. Interestingly, the singlet oxygen generation from DPC was endowed when irradiated at one-photon excitation wavelength of 638 nm and femtosecond 770 nm if considering the strong 2PA capability and large δ2PA values. In addition, DPC showed a photothermal conversion efficiency of up to 36.8% under the excitation NIR-wavelength of 638 nm.
More importantly, inspired by the efficient singlet oxygen generation and reliable photothermal conversion, the excellent phototherapeutic functions of DPC were also investigated using the mouse breast cancer cells (4T1) model. The “three-in-one” phototherapeutic effect of the NIR-wavelength 2PA excitation, photodynamic therapy, and photothermal therapy in the potential photosensitizer DPC was illustrated experimentally and theoretically.
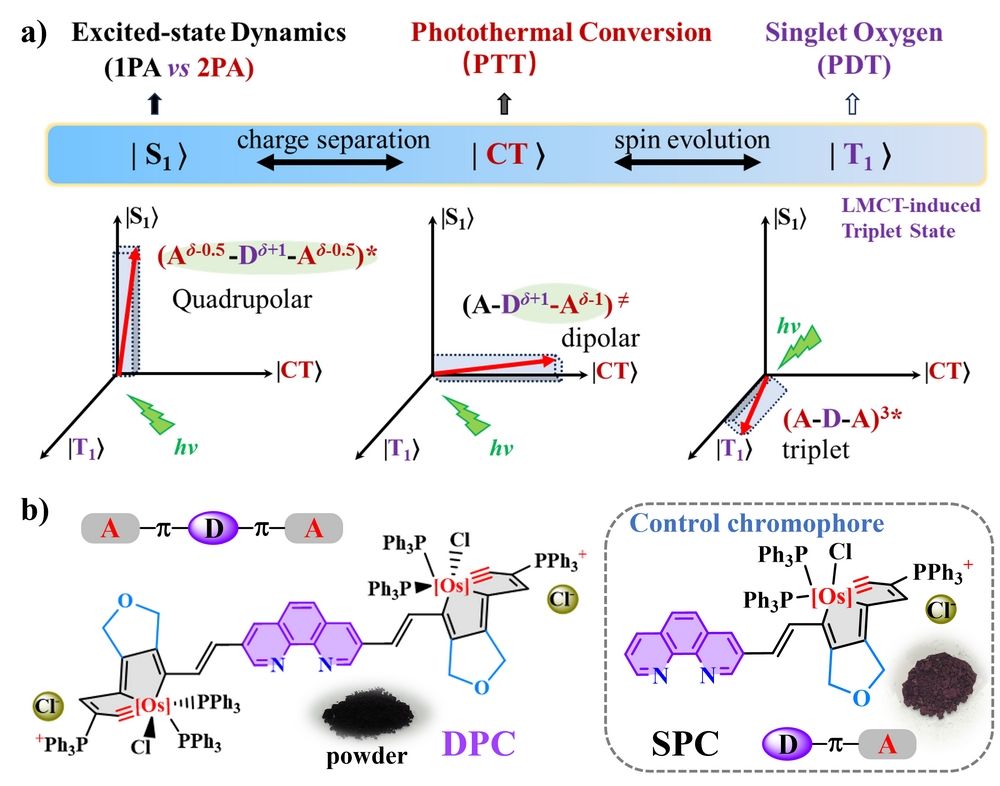
Figure 1. (a) Illustration of the plausible excited-state dynamics of quadrupolar metallaaromatic chromophores and plausible paths for singlet oxygen generation or photothermal conversion; (b) Molecular structures of the double-phenanthroline-carbolong DPC and the control chromophore single-phenanthroline-carbolong SPC.
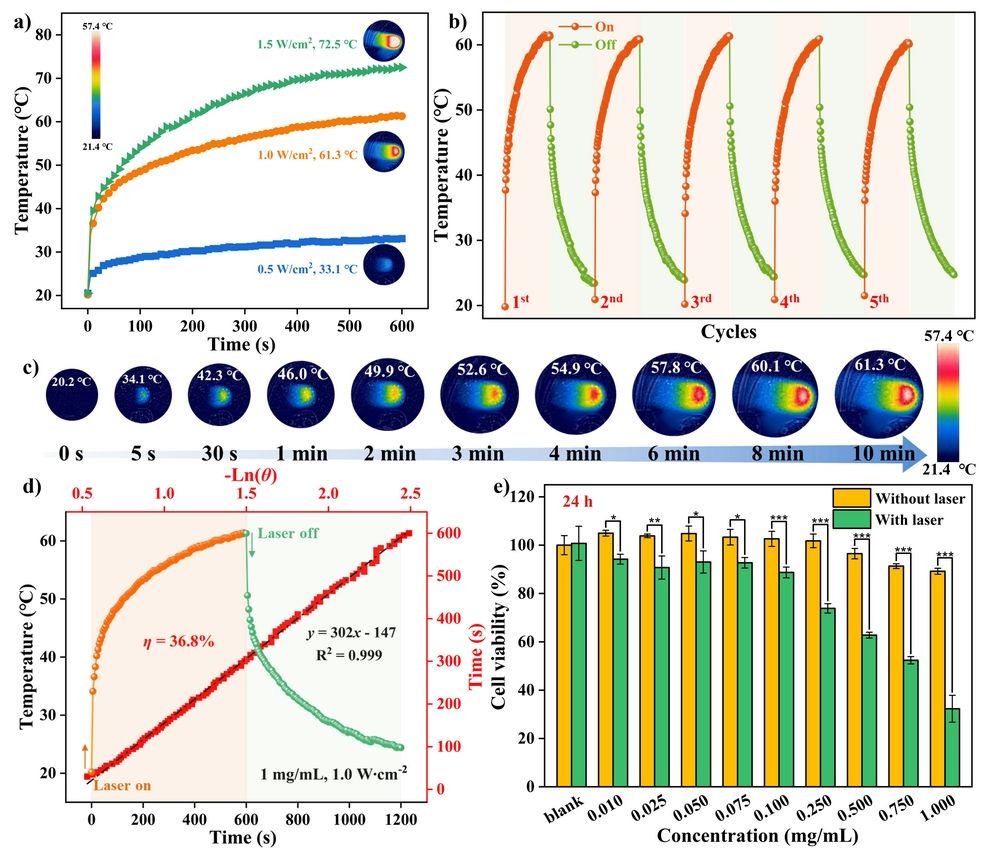
Figure 2. (a) Photothermal heating curves of DPC (660 µL, 1.00 mg/mL) following exposure to 638 nm laser at various power densities; (b) Five photothermal heating curves for DPC under laser on-off irradiation. (c) Infrared thermographies of DPC in water-DMSO solution (9/1, v/v, 1.00 mg/mL, 660 µL) under laser irradiation at various time points; (d) Temperature rise/fall curves and the cooling time vs –ln(ө) plot. Notes: ө represents the driving force temperature for DPC. (e) Cell viability of 4T1 cells incubated with DPC at varying concentrations (0.01 ~ 1.00 mg/mL) for 24 h without or with laser irradiation. Notes: 638 nm laser, 1.0 W/cm2.
First Author: Chang Haixia, Master’s student, Shaanxi Normal University
Correspondence Authors: A/Prof. Liu Taihong and A/Prof. Miao Rong, Shaanxi Normal University
Full Text Link: https://doi.org/10.1039/D5SC00013K