
Yutong Shang, Yalei Ma, Qiangbazhuoma, Baimaquzhen, Liping Ding, Jing Liu, Shiwei Yin, Rong Miao*, and Yu Fang. J. Phys. Chem. Lett. 2025, 16, 3389−3396,DOI: 10.1021/acs.jpclett.5c00774
Fluorescent molecular materials play an indispensable role in sensors, bioimaging and optoelectronic devices because of their excellent photoelectric properties and tunable structural characteristics. However, most dyes lose their fluorescence and exhibit degraded or even quenched emission in solid state, owing to the strong exciton coupling as a result of intermolecular interactions. Unlike fluorescence in solution state, mature theories are lacked in solid state due to the complex influences of molecular structure, molecular conformation, molecular stacking as well as noncovalent interactions, and needs to be systematically studied from the dimension.
Twisted intramolecular charge transfer (TICT) fluorophores are dyes that show drastic conformational change in excited state during the charge transfer process. The excited compound transforms from a quasi-planar conformation to an almost perpendicular conformation through bond rotation, accompanied by fluorescence decrease. The rotation not only affects the intramolecular charge transfer (ICT), but also influences the molecular conformation as well as stacking model in aggregated state. In this work, two TICT fluorophores (Ph-HP and Ph-MP) composed of dimethyl terephthalate and pyrrole/N-methylpyrrole are designed to elucidate the fluorescence mechanism in organic crystalline materials. Based on the unique luminescent properties of TICT fluorophores, this study designed and synthesized two pyrrole derivative-modified single benzene ring TICT fluorophores (Ph-HP and Ph-MP). Through systematic investigation of their photophysical properties in solution state and solid states (including powders and crystals), the physicochemical mechanisms underlying crystallization-induced fluorescence enhancement or quenching were revealed.
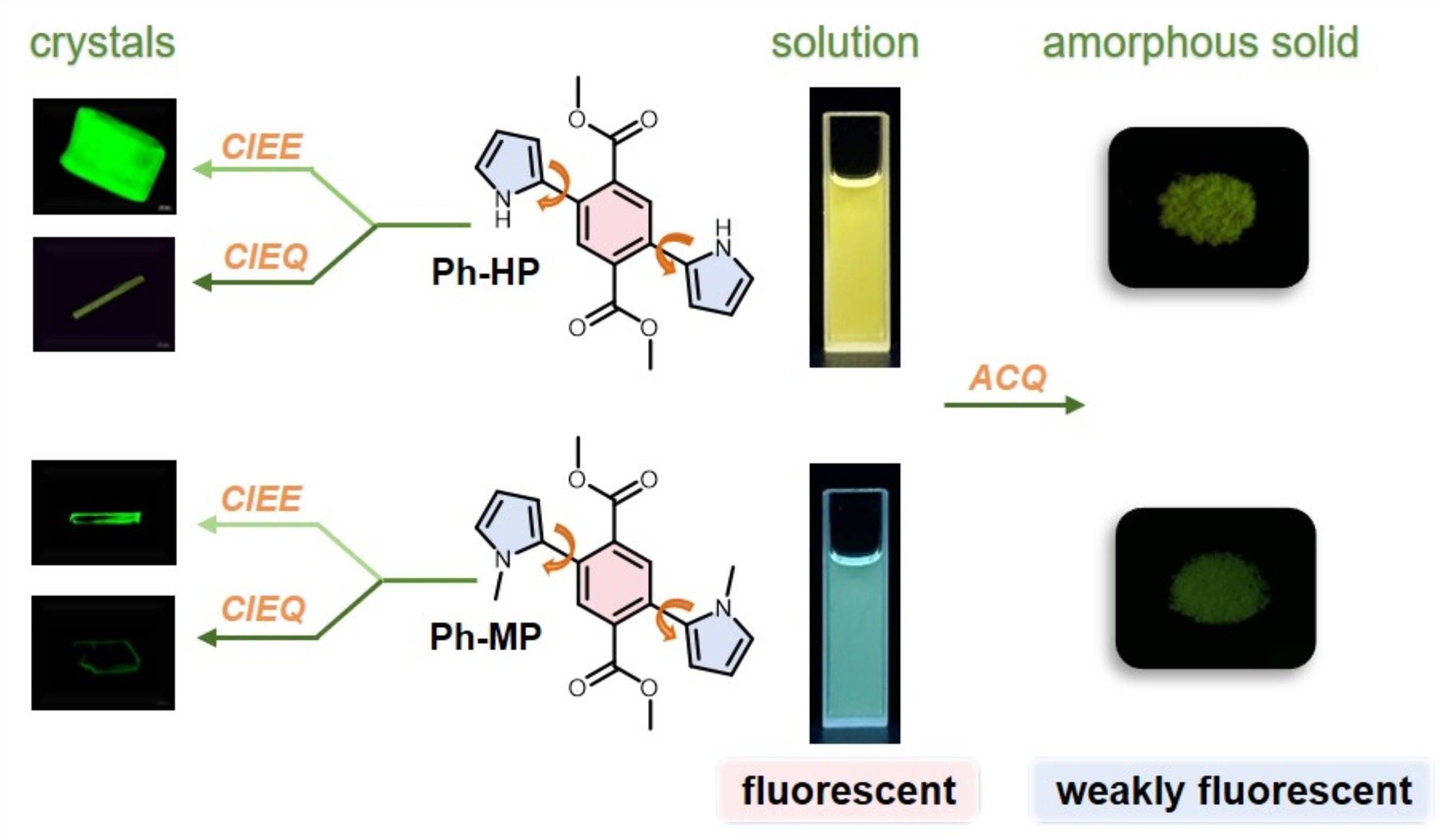
Figure 1. Molecular structures of Ph-MP and Ph-HP, as well as their fluorescence images in solution, powder and crystals.
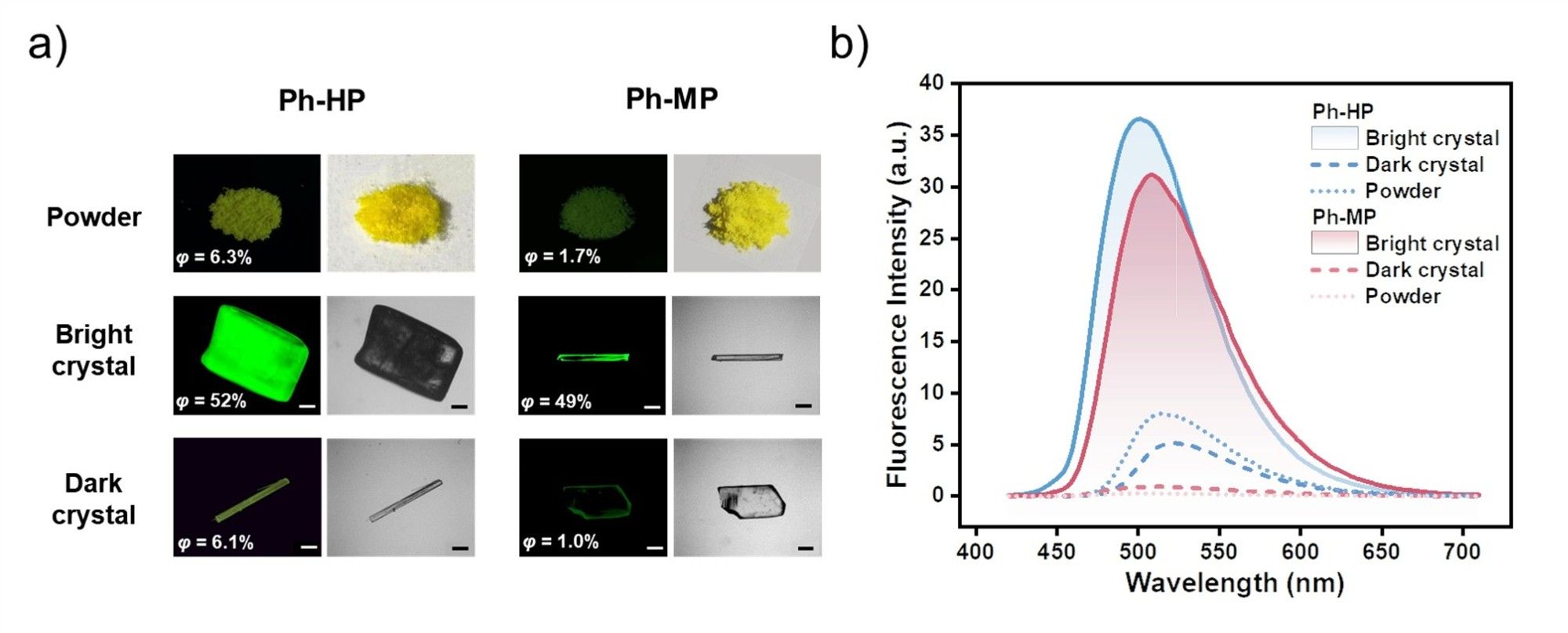
Figure 2. (a) Images of Ph-HP and Ph-MP under UV light and sunlight. (b) Fluorescence emission spectra of Ph-HP and Ph-MP in solid state.
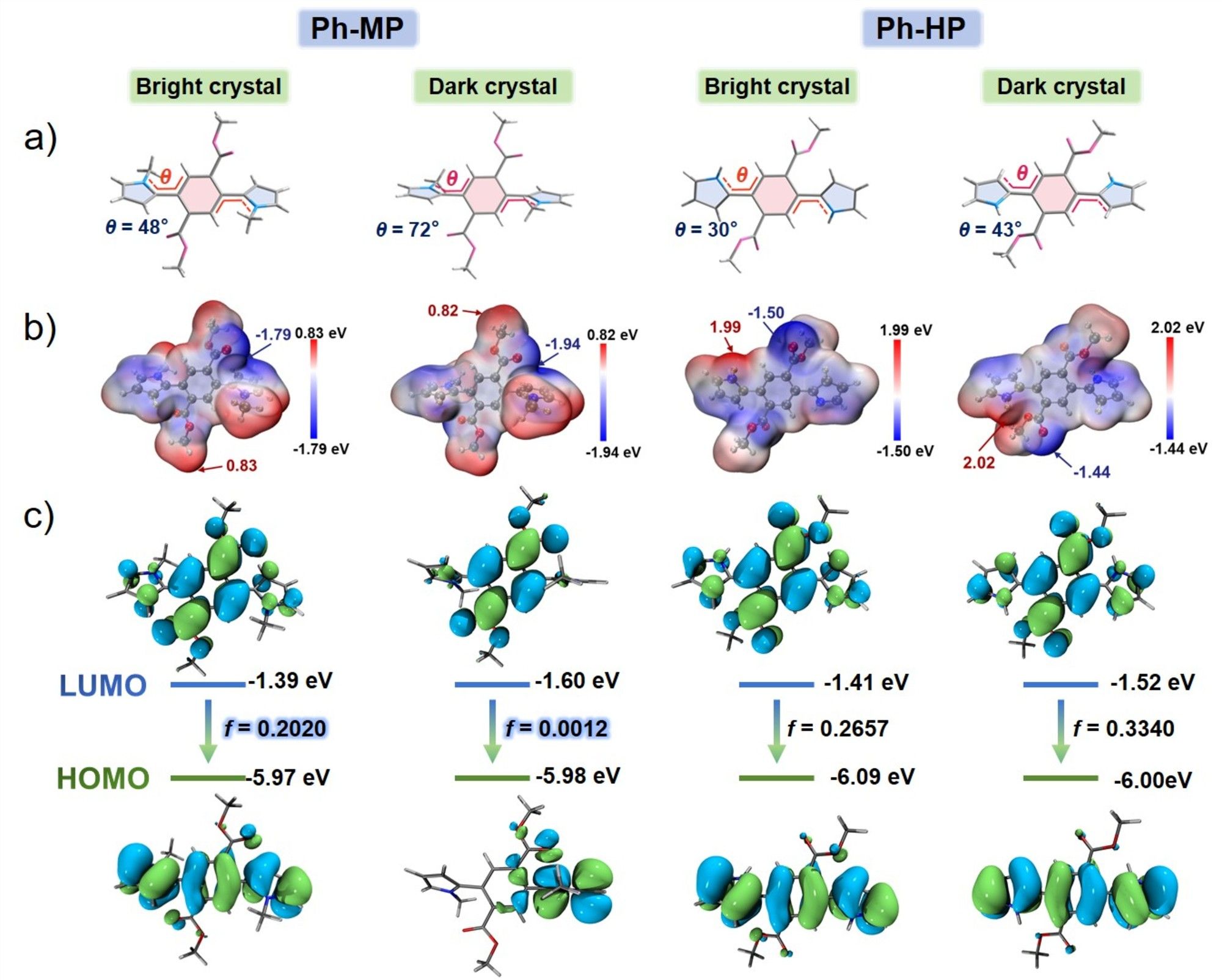
Figure 3. (a) Molecular conformation in the single crystals of Ph-HP and Ph-MP. (b) Molecular electrostatic potential surface and (c) Frontier molecular orbitals of Ph-HP and Ph-MP with different conformations.
Both Ph-HP and Ph-MP exhibit bright fluorescence in toluene solution, yet demonstrate extremely weak fluorescence in their powder states. Interestingly, these two compounds can each form two types of crystals with strikingly distinct fluorescence: one type of crystal exhibits bright fluorescence (Q.Y. > 0.4), while the other shows extremely weak fluorescence (Q.Y. < 0.07). By combining single-crystal analysis results and theoretical calculations, we systematically investigated the effects of molecular conformation, packing arrangements, and intermolecular interactions on crystal fluorescence. The results revealed that although these two molecules are homologues with similar luminescence behaviors, their fluorescence enhancement or quenching in the crystalline state is governed by entirely distinct mechanisms. For Ph-HP, crystal fluorescence is predominantly determined by π-π stacking. Crystals with weaker π-π interactions exhibit fluorescence enhancement compared to the solution state, while those with stronger π-π interactions show fluorescence quenching. In contrast, the luminescence properties of Ph-MP crystals depend on molecular TICT propensity. Crystals formed by molecules with weak TICT propensity exhibit fluorescence enhancement, whereas those with strong TICT propensity demonstrate fluorescence quenching. This study uncovers two distinct mechanisms through which crystallization processes influence fluorescence properties, providing valuable insights for the design and development of novel high-performance luminescent materials.
First Author: Shang Yutong, master’s student, Shaanxi Normal University
Correspondence Author: A/Prof. Miao Rong, Shaanxi Normal University
Full Text Link: https://doi.org/10.1021/acs.jpclett.5c00774