
Yijun Zhang, Yihuan Sun, Huajing Li, Huanhuan Dong, Min Dou, Ting Li, Jing Liu*, Jian Kong, Yuangang Li*. Chem. Eur. J. 2025, e202500804, DOI: org/10.1002/chem.202500804
Chirality refers to the property that an object cannot coincide with its mirror image in space, which is a fundamental characteristic of living matter and nature. All known life forms are homochiral, with DNA and RNA composed of D-sugars and proteins made from L-amino acids. The reason for homochirality in evolution may lie in the preferential selection of specific chiral molecular properties during the origin and evolution of life. Accordingly, studying the chiral competition phenomenon in multiple component system is crucial for understanding the homochirality in nature.
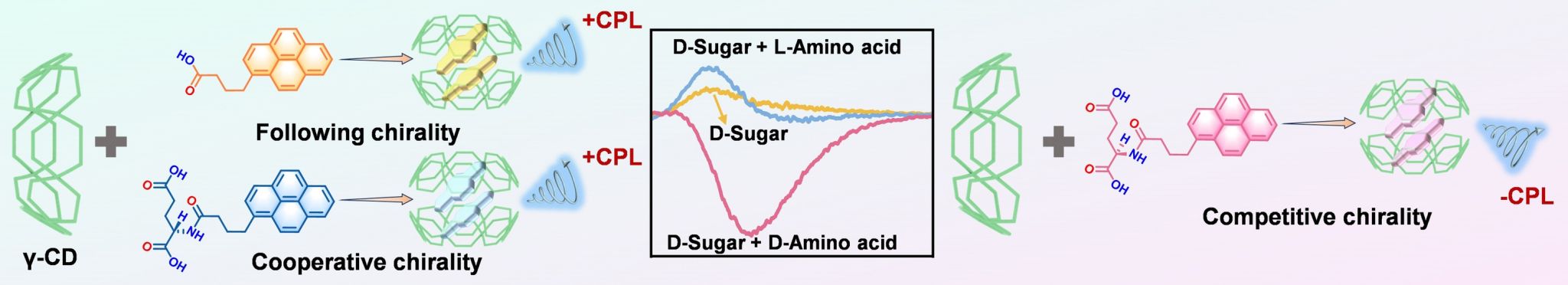
In this study, the chiral competitive effect between carbohydrates and amino acids through the host-guest complex formation was investigated by employing amino acid modified pyrene derivatives and naturally occurring γ-cyclodextrin (γ-CD) as chiral sources, the circularly polarized luminance (CPL) as indicator. Supramolecular complexes of achiral 1-pyrenebutyric acid and L-amino acid-pyrene derivatives with γ-CD exhibited positive CPL signals, with chirality derived from γ-CD’s stereochirality. In contrast, supramolecular complexes formed by D-amino acid-pyrene derivatives with γ-CD showed a negative CPL signal, reflecting chiral competition between D-amino acids and D-glucose in γ-CD.
Fluorescence spectroscopy and ITC measurements show that the chiral pyrene derivatives bind strongly to γ-CD in a 2: 2 ratio, with the pyrene moiety encapsulated in the cavities of two γ-CD molecules in a dimeric form (Figure 1). Mechanism exploration showed that the depth of insertion and stacking arrangement of the guest molecules within the γ-CD cavity played an essential role for the CPL inversion. Interestingly, chiral competition only occurred between D-amino acids and D-carbohydrates, while cooperation was observed between L-amino acids and D-carbohydrates, which were consistent with the natural chirality bias. This work not only advances our understanding of chiral competition and cooperation in multicomponent systems but also provides an inspiring starting point for exploring chirality regulation in chiral multicomponent systems, with potential applications in molecular recognition, sensing and other fields.
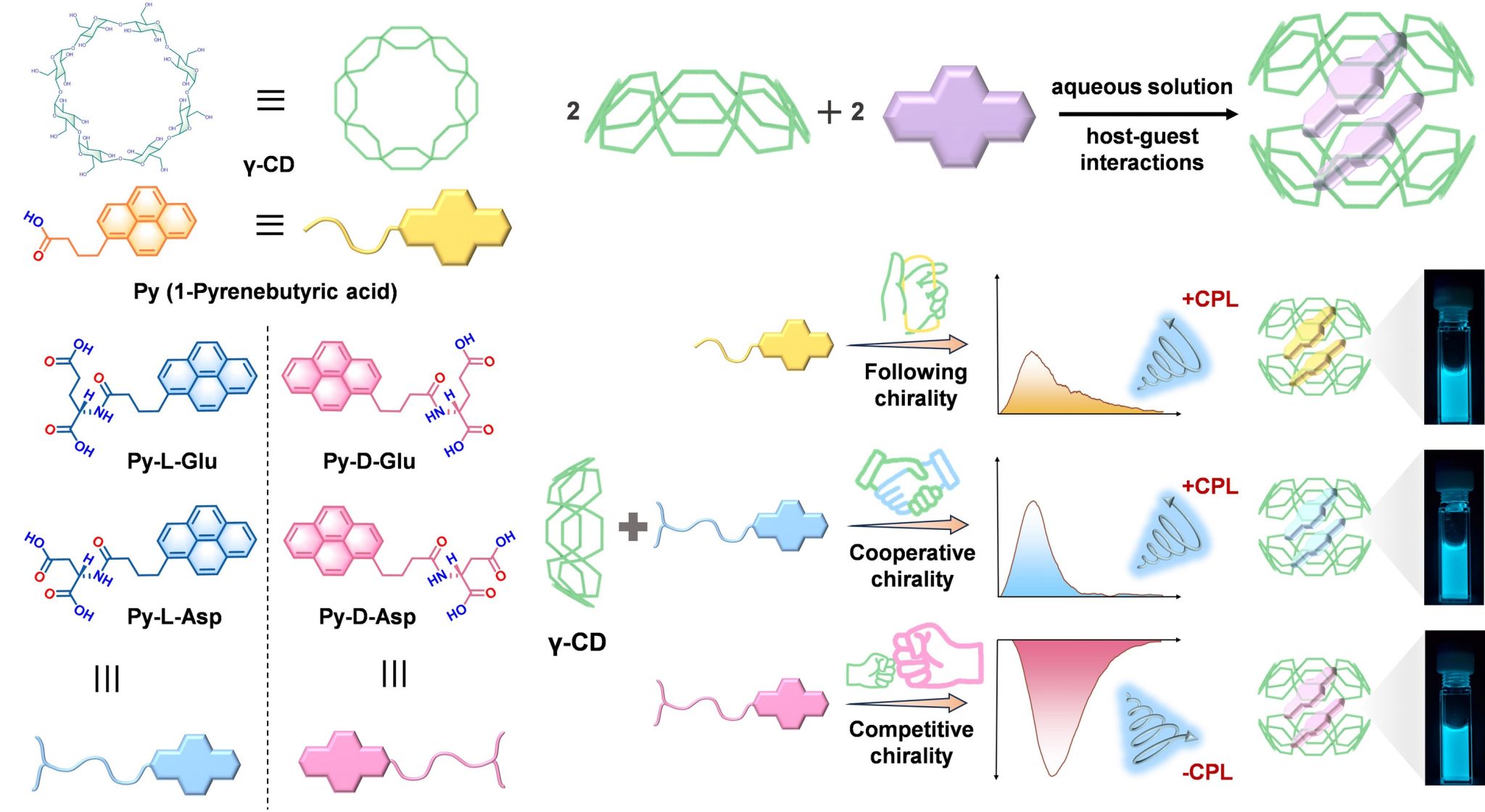
Figure 1. The schematic diagram of the binding of different pyrene derivatives to γ-CD molecules to regulate the supramolecular chirality.
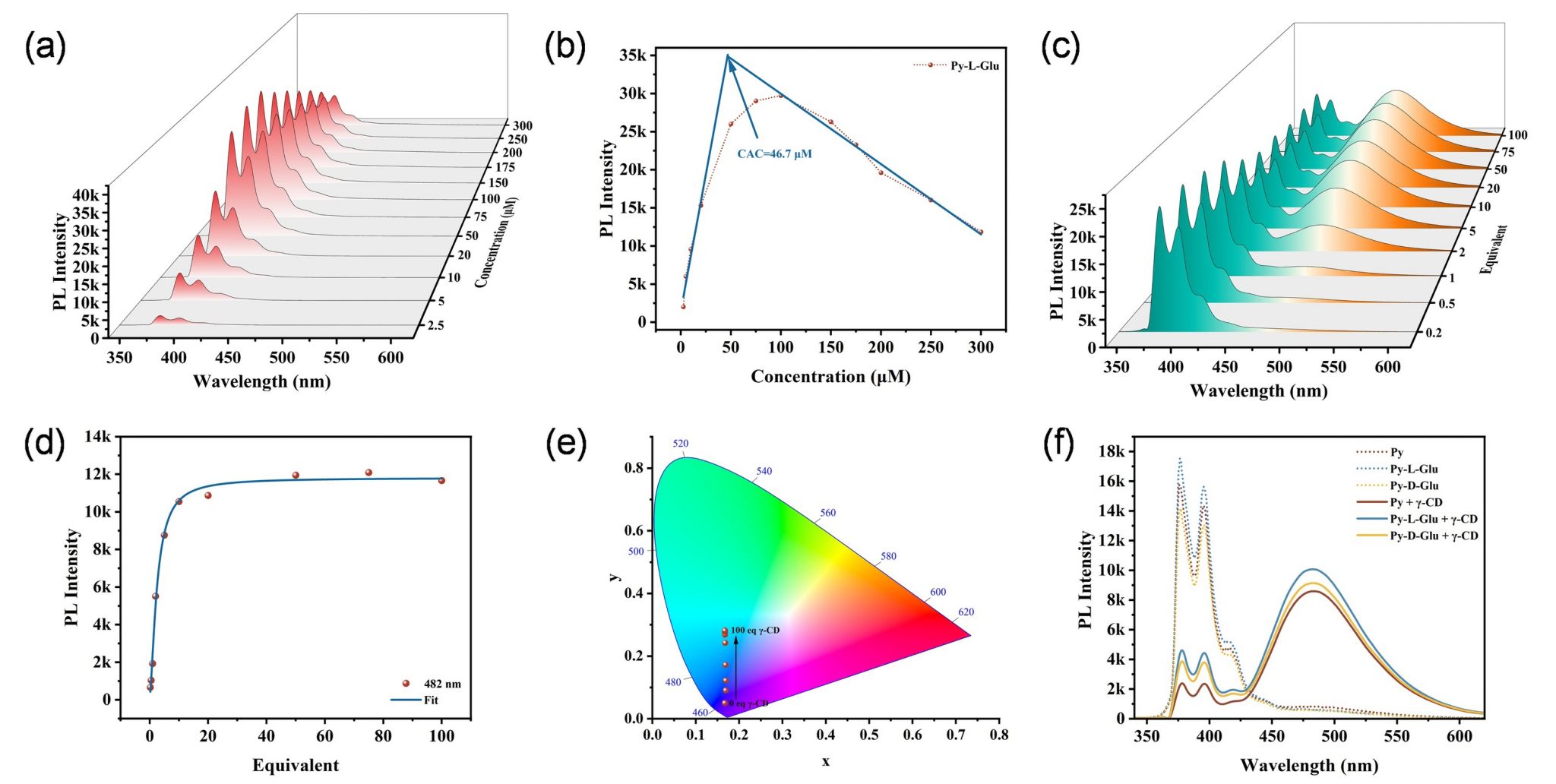
Figure 2. The binding of different pyrene derivatives to γ-CD molecules to regulate the fluorescence emission properties.
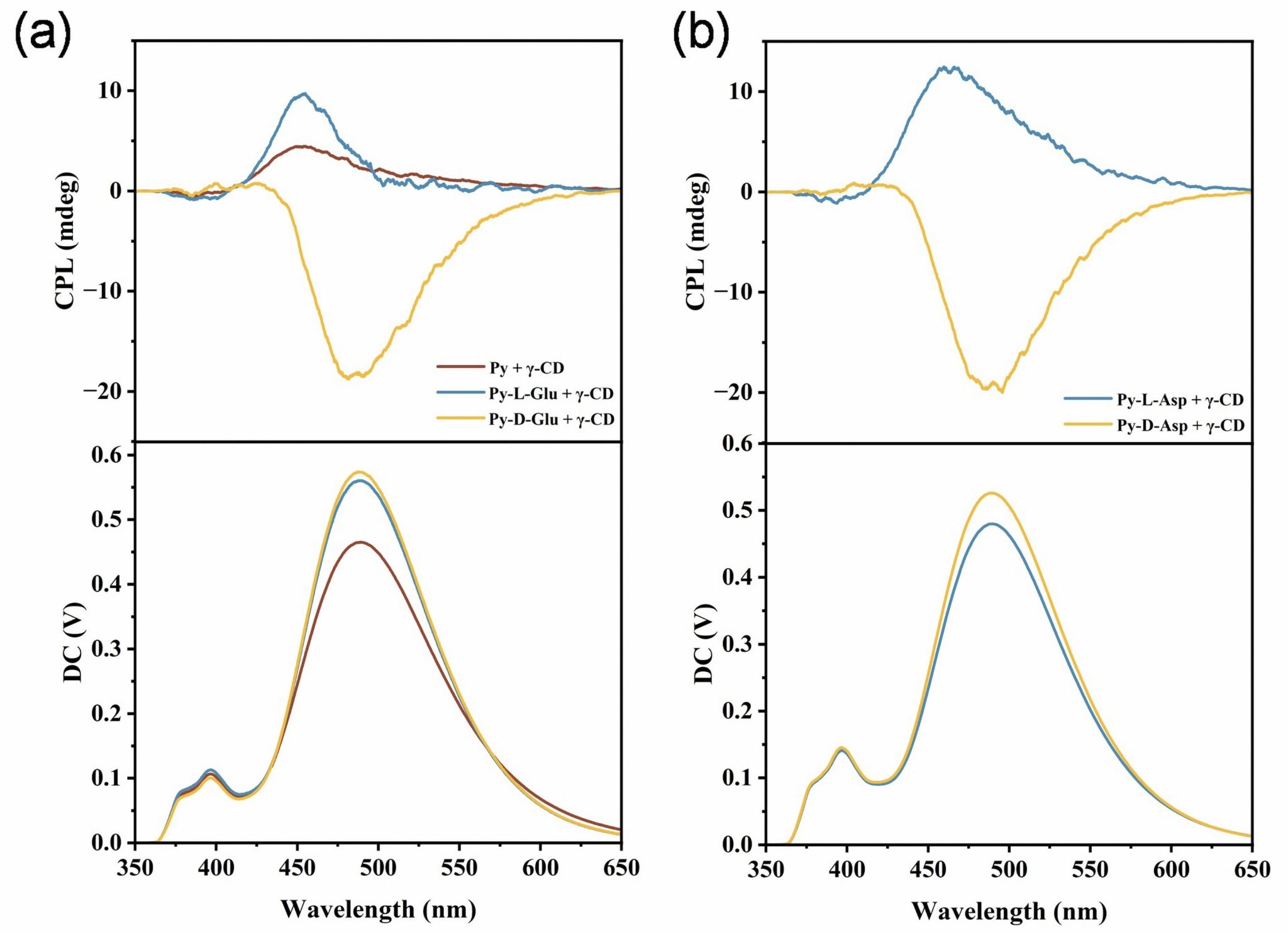
Figure 3. The binding of different pyrene derivatives to γ-CD molecules to regulate the CPL properties.
First Author: Zhang Yijun, master’s student, Xi’an University of Science and Technology
Correspondence Authors: Prof. Li Yuangang, Xi’an University of Science and Technology; Prof. Liu Jing, Shaanxi Normal University
Full Text Link: https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/chem.202500804